Achieving Medical-Grade Accuracy: A Case Study with SLA Printers
Medical device manufacturing demands precision that exceeds conventional manufacturing tolerances. SLA printer technology has emerged as a transformative solution for producing medical-grade components with exceptional accuracy and reliability. Through controlled photopolymerization processes, these systems achieve dimensional tolerances within ±0.1mm, enabling manufacturers to produce surgical guides, dental models, and biocompatible prototypes that meet stringent FDA and ISO 13485 standards. This comprehensive case study examines how advanced stereolithography systems revolutionize medical manufacturing workflows while addressing critical procurement considerations for B2B decision-makers.
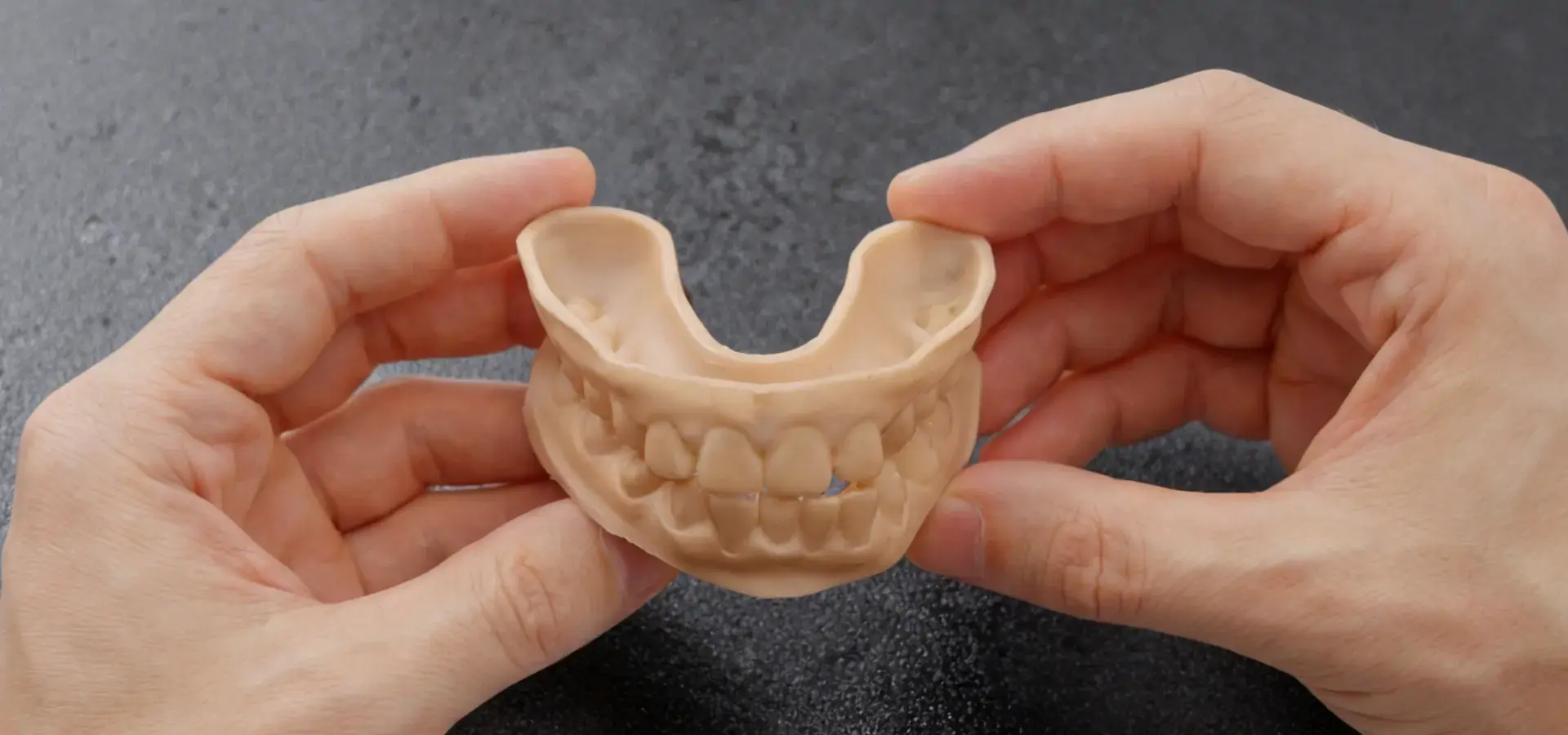
Understanding SLA Printer Technology and Its Role in Medical-Grade Accuracy
Stereolithography represents the pinnacle of additive manufacturing precision, utilizing UV lasers to cure liquid photopolymer resins layer by layer. This fundamental process creates parts with surface finishes approaching injection-molded quality while maintaining dimensional accuracy critical for medical applications.
Technical Specifications That Drive Medical-Grade Performance
The SLA printer characteristics of mechanical SLA frameworks specifically affect their reasonableness for restorative device generation. Determination capabilities ordinarily run from 25 to 100 microns, with layer statures movable between 0.01mm and 0.15mm depending on application requirements. Advanced frameworks consolidate German Scanlab galvanometers and AOC laser arrays that provide reliable bar quality over the whole construct platform. Variable spot-size laser innovation speaks to a critical advancement in adjusting speed with accuracy. Expansive laser spots of 0.5-0.6mm quicken inner filling operations, whereas fine spots of 0.18-0.2mm guarantee exact form definition and complicated detail propagation. This dual-approach strategy increments printing speeds by 30-50% compared to ordinary single-spot frameworks without compromising dimensional accuracy.

Biocompatible Material Compatibility and Regulatory Compliance
Medical-grade photopolymer gums experience thorough testing to accomplish USP Lesson VI certification and ISO 10993 biocompatibility benchmarks. These materials show great mechanical properties counting pliable qualities surpassing 50 MPa and stretching rates appropriate for adaptable restorative components. Open-source gear plans empower producers to select gums from numerous providers, giving fetched adaptability whereas keeping up quality standards. The integration of profound learning calculations optimizes printing parameters naturally, lessening human mistakes and guaranteeing reliable portion quality over generation runs. These shrewd frameworks analyze geometric complexity and alter introduction times, laser control, and checking designs to accomplish ideal curing throughout each layer.
Challenges in Achieving Medical-Grade Accuracy with SLA Printers
Despite technological advances, several obstacles can compromise medical-grade quality if not properly addressed through systematic quality management approaches.
Common Print Defects and Quality Control Measures
Calibration mistakes speak to the most common source of dimensional mistakes in SLA generation. Tar temperature varieties, dishonorable stage leveling, and laser control irregularities can present dimensional deviations surpassing worthy therapeutic resiliences. Actualizing comprehensive calibration conventions, including exactness estimation apparatuses and factual handle control strategies, guarantees reliable yield quality. Print bolster optimization poses another basic challenge, especially for complex restorative geometries with overhangs and empty areas. Lacking bolster structures can cause warpage or surface surrenders, whereas intemperate underpins may harm sensitive highlights during evacuation. Progressed cutting program with computerized support era calculations minimizes these issues, while protecting surface integrity.
Material Handling and Environmental Considerations
Photopolymer gums show sensitivity to temperature fluctuations and the introduction of surrounding light. Capacity temperatures must stay inside the 18-25°C range to avoid thickness changes that influence layer grip and curing consistency. Controlled fabricating situations with steady mugginess levels between 40-60% avoid condensation issues that may compromise print quality. Post-processing workflows require careful consideration of washing solvents and UV curing parameters. Inadequate dissolvable expulsion can take off shabby buildups that draw in contaminants, whereas over-curing may present brittleness or dimensional changes. Standardized post-processing conventions with reported timing and temperature parameters guarantee reproducible comes about over generation batches.
Case Study: Implementing SLA Printers for Medical Device Production
A leading medical device manufacturer recently integrated industrial-grade stereolithography systems to produce customized surgical guides and dental prosthetics, demonstrating the practical implementation of medical-grade SLA technology.

Project Requirements and Equipment Selection
The extent requested dimensional precision inside ±0.05mm for surgical direct applications, with surface wraps up appropriate for coordinate understanding contact. Construct volume necessities included single-piece generation of components up to 200mm in length, requiring large-format SLA capabilities. Fabric compatibility necessities enveloped both inflexible biocompatible gums for auxiliary components and adaptable materials for understanding comfort applications. Equipment assessment centered on frameworks, including marble base development for improved stability and Panasonic AC servo engine drives for exact situating control. The chosen arrangement joined German-engineered galvanometer frameworks able of keeping up spot accuracy over 300mm construct stages, while supporting variable spot-size operations for optimized throughput.
Workflow Implementation and Quality Validation
The generation workflow consolidated plan approval conventions utilizing 3D checking confirmation to affirm CAD demonstrate precision, some time recently printing. Mechanized bolster era calculations decreased manual arrangement time by 60% whereas keeping up reliable bolster arrangement for ideal portion quality. Real-time observing frameworks followed the temperature, laser control yield, and natural conditions throughout each construct cycle. Quality approval utilized a measuring machine (CMM) assessment conventions to confirm dimensional compliance over basic highlights. Measurable investigation of 500 successive parts illustrated dimensional consistency within ±0.03mm ranges, surpassing the starting exactness requirements. Surface unpleasantness estimations found the middle value of Ra 1.2 microns, reasonable for coordinate therapeutic application without extra wrapping up operations.
Performance Results and Production Metrics
The usage of SLA printer execution changes compared to conventional fabricating strategies. Generation lead times diminished from 2-3 weeks to 24-48 hours for custom surgical guides, empowering same-day persistent methods in crisis circumstances. Fabric squander diminishment surpassed 85% compared to subtractive fabricating approaches, whereas labor costs per portion diminished by 40% through computerized generation workflows. Failure rate examination uncovered print victory rates over 98% after beginning handle optimization, with remaining disappointments ascribed to client mistake or hardware restrictions. Persistent operation testing illustrated steady execution over 1000+ hour generation runs without noteworthy corruption in yield quality or dimensional accuracy.
Procurement Insights: Selecting and Buying the Right SLA Printer for Medical Applications
Strategic procurement decisions require a comprehensive evaluation of technical capabilities, total cost of ownership, and supplier support infrastructure to ensure long-term operational success.
Market Analysis and Vendor Comparison
The mechanical SLA advertisement highlights a few established producers advertising medical-grade capabilities, each with unmistakable preferences for particular applications. Frameworks consolidating worldwide component brands counting German galvanometers and accuracy laser gatherings ordinarily illustrate prevalent unwavering quality and precision compared to cost-optimized alternatives. Build volume capabilities extend from desktop units, reasonable for dental applications, to large-format frameworks obliging orthopedic embed models. Entry-level mechanical units offer 150x150x200mm construct envelopes satisfactory for most dental and little restorative component generation, whereas progressed frameworks give 600x600x400mm volumes for clump generation or huge prosthetic devices.
Total Cost of Ownership Considerations
Initial hardware speculation speaks to, as it were, 30-40% of five-year operational costs for therapeutic SLA generation. Consumable costs counting photopolymer gums and substitution components, essentially affect long-term productivity. Open-source fabric compatibility has taken a toll on focal points by empowering competitive fabric sourcing while keeping up quality standards. Maintenance prerequisites shift essentially between producers, with a few frameworks requiring specialized service contracts, whereas others rely on in-house support conventions. Prescient support capabilities utilizing IoT sensors and inaccessible observing frameworks decrease spontaneous downtime, while optimizing component substitution plans based on real utilization designs rather than subjective time intervals.
Support Infrastructure and Service Capabilities
Technical bolster responsiveness fundamentally impacts generation coherence, especially for time-sensitive therapeutic applications. Driving providers offer 24/7 accessible symptomatic capabilities with ensured reaction times beneath one hour for basic issues. On-site benefit accessibility inside 4-hour windows guarantees negligible generation disturbance amid hardware malfunctions. Training programs encompassing hardware operation, fabric handling, and quality control methods quicken administrator capability, while diminishing user-induced disappointments. Comprehensive documentation counting standard working strategies and investigation guides, underpins reliable operation over numerous shifts and administrator changes.
Magforms: Advanced SLA Solutions for Medical Manufacturing
Magforms specializes in delivering cutting-edge stereolithography solutions engineered specifically for medical-grade applications. Our product portfolio combines high-precision industrial SLA systems with certified biocompatible materials to support compliant medical device manufacturing across multiple specialties, including dentistry, orthopedics, and surgical instrumentation.

Superior Hardware Engineering and Performance Specifications
Our industrial SLA printer incorporates premium international components, including German Scanlab galvanometer assemblies, AOC laser systems, and Panasonic AC servo motor drives to ensure exceptional performance and long-term reliability. The flagship Helios-P450 model features marble base construction that achieves printing accuracy of ±0.1mm (L ≤ 100mm), delivering near industrial-grade stability suitable for the most demanding medical applications. Large-format printing capabilities enable single-build production of oversized functional components or efficient batch processing of multiple medical devices simultaneously. This scalability breaks traditional size limitations while maintaining consistent quality across the entire build platform, supporting both prototype development and small-volume production requirements.
Innovative Technology for Enhanced Productivity
Variable spot-size laser technology represents our core technological advancement, delivering 30-50% speed improvements compared to conventional printing methods. Intelligent algorithms automatically select optimal laser parameters, utilizing large spots for rapid internal filling and fine spots for precise contour definition and detailed features. Deep learning integration provides additional performance optimization, achieving further 20% speed improvements through predictive scanning path optimization. These innovations address the longstanding industry challenge of balancing high efficiency with precision requirements, enabling medical manufacturers to meet tight production schedules without compromising quality standards.
Flexible Design and Material Compatibility
Open-source equipment architecture allows customers complete freedom in photopolymer resin selection, breaking proprietary material limitations that restrict cost control and operational flexibility. This approach enables medical manufacturers to source certified biocompatible materials from preferred suppliers while maintaining optimal printing performance. Our product range spans desktop-level systems suitable for dental practices to industrial-grade units supporting scaled manufacturing operations. This comprehensive portfolio ensures appropriate solutions for small studios requiring occasional custom work as well as large enterprises implementing volume production workflows.
Conclusion
Medical-grade SLA printer represents a transformative manufacturing technology that enables unprecedented precision and customization in medical device production. Through advanced laser control systems, optimized material compatibility, and intelligent process automation, modern stereolithography achieves dimensional tolerances and surface finishes that meet the most stringent medical standards. Successful implementation requires careful attention to equipment selection, workflow optimization, and quality control protocols, but the resulting benefits include dramatically reduced lead times, enhanced customization capabilities, and improved patient outcomes through precisely fitted medical devices.
FAQ
What specific accuracy levels can medical-grade SLA systems achieve?
Industrial SLA systems equipped with precision components can achieve dimensional accuracy within ±0.1mm for parts under 100mm in length, with some advanced configurations reaching ±0.05mm tolerances. These accuracy levels meet FDA requirements for most medical device categories, including surgical guides and dental prosthetics.
How do variable spot-size technologies improve medical printing applications?
Variable spot-size laser systems automatically adjust beam diameter based on geometric requirements, using large spots (0.5-0.6mm) for rapid internal filling and fine spots (0.18-0.2mm) for detailed contours. This approach increases printing speeds by 30-50% while maintaining precision requirements for medical applications.
What biocompatible materials are available for medical SLA printing?
Medical-grade photopolymer resins include USP Class VI certified materials suitable for temporary patient contact, along with specialized formulations for dental applications meeting ISO 10993 biocompatibility standards. These materials offer mechanical properties, including tensile strengths exceeding 50 MPa and controlled flexibility for various medical applications.
How important is post-processing for achieving medical-grade quality?
Post-processing protocols critically impact final part quality, requiring controlled washing procedures and precise UV curing parameters. Standardized workflows ensure complete solvent removal and optimal material properties while preventing over-curing that could compromise dimensional accuracy or introduce brittleness.
What support infrastructure should be evaluated when selecting SLA printer suppliers?
Essential support considerations include 24/7 technical assistance with guaranteed response times, comprehensive training programs for operators, predictive maintenance capabilities, and documented standard operating procedures. Reliable support infrastructure ensures consistent production continuity for time-sensitive medical applications.
Partner with Magforms for Medical-Grade SLA Solutions
Medical device manufacturers seeking reliable, high-precision stereolithography solutions will find that Magforms offers unmatched expertise and advanced technology tailored for demanding healthcare applications. Our industrial SLA printer systems combine German-engineered galvanometers, premium laser assemblies, and intelligent processing algorithms to deliver medical-grade accuracy with enhanced productivity.
Magforms serves over 300 enterprises globally with comprehensive solutions encompassing precision equipment, certified biocompatible materials, and complete technical support. Our open-source design philosophy provides material sourcing flexibility while maintaining strict quality standards essential for medical compliance.
Contact our technical specialists at info@magforms.com to discuss your specific medical manufacturing requirements and explore customized SLA printer supplier solutions that accelerate your production capabilities while ensuring consistent medical-grade quality.
References
1. Gibson, Ian, et al. "Additive Manufacturing Technologies: 3D Printing, Rapid Prototyping, and Direct Digital Manufacturing." Springer Science & Business Media, 2015.
2. Melchels, Ferry P.W., et al. "A Review on Stereolithography and Its Applications in Biomedical Engineering." Biomaterials Journal, Vol. 31, 2010.
3. Stansbury, Jeffrey W., and Mike J. Idacavage. "3D Printing with Polymers: Challenges among Expanding Options and Opportunities." Dental Materials, Vol. 32, 2016.
4. Ventola, C. Lee. "Medical Applications for 3D Printing: Current and Projected Uses." Pharmacy and Therapeutics, Vol. 39, 2014.
5. Zhai, Xiaojun, et al. "Additive Manufacturing: Making Imagination the Major Limitation." JOM Journal of the Minerals, Metals and Materials Society, Vol. 66, 2014.
6. Morrison, Robert J., et al. "Regulatory Considerations in the Design and Manufacturing of Implantable 3D-Printed Medical Devices." Clinical and Translational Science, Vol. 8, 2015.


Magforms makes design and manufacture easier.
Popular Blogs
-
 Industry InsightsManufacturing Industry
Industry InsightsManufacturing IndustryFDM vs. SLA Printer: Which Technology Is Right for Your Project?
-
 Industry InsightsManufacturing Industry
Industry InsightsManufacturing IndustryTop 5 Applications of Industrial 3D Printers in Manufacturing Today
-
 Industry InsightsManufacturing Industry
Industry InsightsManufacturing IndustryWhy Stereolithography 3D Printers Are Unmatched for Microfluidic Device Prototyping?
