Large Scale Resin 3D Printer for Dental and Medical Models
When dentistry labs and companies that make medical devices need to make a lot of them, they need a large-scale resin 3D printer. These industrial systems use vat photopolymerization technologies such as SLA, DLP, and mSLA to selectively cure liquid photopolymer resins layer by layer with high dimensional accuracy. This makes it possible to make anatomical models, surgery guides, and orthodontic devices that are capable of achieving dimensional tolerances within tens of microns under validated production conditions. Unlike desktop printers, industrial-grade large format machines can print larger objects—often over 300 mm on multiple axes—so they can make dozens of dental aligners or full-scale anatomical replicas at the same time, meeting the strict needs of healthcare manufacturing.
What is a large-scale resin 3D printer, and how does it work?
A large-scale resin 3D printer brings together precision optics and industrial engineering. It was made for high-throughput production settings in the medical and dental fields. Vat photopolymerization is the core operating principle behind these industrial SLA systems. A UV or visible light source carefully hardens liquid resin into layers. In industrial SLA printing, “large-scale” typically refers to systems offering build volumes exceeding approximately 300 mm on at least one axis, enabling batch production of dental and medical components. This means that full dental arches, life-size head models, or batches of surgery templates can be printed without any problems.
Core Technologies Behind Medical-Grade Resin Printing
Industrial resin systems are powered by three main technologies. Stereolithography (SLA) uses precise laser beams to trace the shape of each layer. This method is well suited for producing complex geometries with fine feature resolution and layer heights commonly ranging from 25 to 100 microns. Digital Light Processing (DLP) uses digital micromirror arrays to cast whole layers at once, which accelerates the production process. Masked Stereolithography uses LCD masking technology combined with LED light sources to expose entire layers simultaneously, providing a balance between throughput, resolution, and equipment cost. Modern medical-grade systems use lasers with varying spot sizes and optimized scanning strategies and intelligent exposure control to improve throughput efficiency while maintaining dimensional consistency compared to older units while maintaining repeatable dimensional accuracy across the validated build area.

Essential Components Ensuring Precision and Repeatability
The base is the resin vat, which is made of inert materials like fluoropolymer-based release films such as FEP, PFA, or ACF materials to keep photopolymers safe from breaking it down chemically. Modern vat systems have two tanks, which allow resin to be automatically replenished during long production runs. The light source, like galvanometer-steered lasers or collimated LED arrays, needs to provide even illumination across the whole platform. Industrial SLA optical systems are designed to maintain uniform light distribution across the build platform. Build platforms have quick-release build platform systems and automated platform calibration systems that get rid of the need to recalibrate between jobs. This is crucial for medical manufacturing environments operating under FDA-regulated quality systems that require repeatable batch consistency.
Specialized Resin Materials for Healthcare Applications
In medical and dental settings, biocompatible plastics that meet ISO 10993 and USP Class VI guidelines are needed. After curing, dental model resins don't change much in size, so they keep the standards needed for crown fitting and implant placement. Surgical guide materials are strong so that they can be drilled accurately and can also withstand up to 134°C autoclave cycles of cleaning. Transparent biocompatible resins support visualization of internal anatomical structures, and flexible materials can mimic the qualities of soft tissue for practicing surgery. Its mechanical qualities range from a tensile modulus of 2,500 MPa for rigid uses to shore hardness values comparable to gingival tissue. The chemicals that make up the material are usually methacrylate or epoxy-based oligomers with photoinitiators.
Industrial SLA systems designed for medical manufacturing often use heated resin tanks and temperature-controlled build chambers that keep the viscosity of the resin at the best flow rates, which are between 25°C and 30°C. This thermal control improves resin flow behavior and helps maintain reliable interlayer curing performance. This environmental control is essential when working with high-temperature resins that can survive steam sterilization routines or ceramic-filled materials intended for high-strength modeling and non-implantable engineering applications.
Benefits of Using Large Scale Resin 3D Printers for Dental and Medical Models
Healthcare increasingly relies on industrial resin printing due to its ability to enhance production efficiency, improve part quality, and yield better patient outcomes. These methods address some of the most significant issues with traditional manufacturing and enable personalized care.
Superior Precision and Surface Quality
Resin-based additive manufacturing can make layers that are 25 to 50 microns thick, which lets it reproduce fine anatomical geometries that are difficult to achieve with lower-resolution extrusion-based technologies. This resolution accurately reproduces fine anatomical details such as interdental papillae and gingival margins from intraoral scan data. Surgical cutting guides made at this level allow osteotomies to be accurate to within 0.5 mm, which lowers the risk of complications and raises the success rate of implant placement, as shown in clinical research.The naturally smooth surface finish of SLA-printed parts, which is significantly smoother than FDM processes, often reduces post-processing requirements. This speeds up the time it takes to deliver parts for time-sensitive surgery planning.
Accelerated Production Cycles and Scalability
Large-format SLA systems with extended XY build dimensions allow multiple cases to be nested simultaneously. With a large-scale resin 3D printer, you can make 40 dental study models or 15 surgery guides in a single overnight print cycle. This means that you can now do in-house overnight operations that used to take weeks to outsource. This parallel production ability can be scaled up without linear time increases—batch production efficiency improves significantly because multiple parts can often be produced within similar layer exposure cycles, which changes the costs of production in a fundamental way. Laboratories say that response times have gone down from 7–10 business days to 24–48 hours, which has a direct effect on how happy patients are and how well clinical scheduling works.
Cost-Effectiveness Through Material Optimization
When compared to subtractive production, vat photopolymerization makes better use of materials. The resin used equals the part volume plus the minimum support structures. Any extra resin that is filtered out can be used again. This means that 85–90% of the material is used when making batches of orthodontic models, compared to 40–50% when cut blanks are used. Because injection molding doesn't need expensive tools, it's possible to make small amounts of something; the number of units needed to break even drops from thousands to tens. When switching between different types of resin, industrial systems with two tanks reduce the amount of waste. This is especially important when keeping stock of specialized safe formulas.
Versatility Across the Medical Product Lifecycle
In healthcare companies, these manufacturing tools do more than one thing. Research teams use these manufacturing tools to ensure the accuracy of physical models during device production. Clinical teams use CT and MRI data to make surgery guides that are specific to each patient. Under the right quality control methods, production sites make Class I and Class II medical devices for final use. Using a single platform to make different types of resin models, such as rigid diagnostic models and flexible soft tissue simulations, cuts down on the need for expensive tools and floor space compared to keeping separate manufacturing technologies.
The photopolymerized parts generally exhibit more uniform mechanical properties than FDM components of photopolymerization ensure that the strength is the same in all directions. This is different from FDM parts, which are weak in different places and easily delaminate. This structural stability is crucial for surgery guides that have to withstand drilling forces and anatomical models that have to be handled many times during case planning sessions.

Comparison and Selection Criteria: Large Scale Resin 3D Printers vs. Other Technologies
When B2B buying teams are looking at investments in additive manufacturing, they need to know how industrial large-scale resin 3D printer systems stack up against other methods and what makes high-end options different from low-cost ones.
Resin Photopolymerization vs. Fused Deposition Modeling
With FDM technology, thermoplastic strands are deposited one layer at a time, allowing for build amounts similar to those of large-scale resin 3D printer systems but with lower initial costs. Resolution and surface clarity, on the other hand, are what make them different. FDM layer heights are usually between 100 and 300 microns, and for medical uses, the obvious layer lines need a lot of post-processing. Because deposited strands are not all the same, they create mechanical weak spots along the edges of layers. This is a problem for surgery guides that have to withstand forces acting in more than one way. Resin systems provide injection-mold surface finishes right off the build platform with more uniform mechanical properties compared with FDM components, so they can meet the strict requirements for medical devices without the need for extra steps. Another difference is that medical-grade FDM filaments are available, but the photopolymer ecosystem has a wider range of validated biocompatible resin formulations designed for regulated dental and medical workflows that are specially designed for short-term tissue contact inside the mouth.
Industrial-Grade vs. Desktop Resin Units
It's clear that the build rate is what sets one company apart from another, but business factors are just as important. Desktop units (platforms smaller than 200 mm) need a lot of human work, which makes it hard to leave them alone and work. Automated resin management, remote tracking, and job queuing systems are built into industrial tools, which makes it possible to support lights-out manufacturing workflows with minimal operator intervention. Print reliability is another major differentiator between industrial and desktop systems. Commercial systems have >95% first-pass yield thanks to improved peel force control using ACF films or tilting vat mechanisms, while desktop units often fail to stick to big cross-sections. Temperature-controlled rooms keep prints consistent across a range of environmental conditions. This capability is important for making medical devices that meet ISO 13485 quality standards and require environmental paperwork.
Key Features Defining 2026 Premium Systems
Leading platforms use several technologies that set them apart from standard options. Variable spot-size laser systems change the beam width on the fly, which speeds up areas that need to be filled in faster while keeping the accuracy of small features—something that fixed-beam designs can't do. Advanced slicing software with AI-driven support generation cuts down on post-processing work and makes sure that parts are oriented correctly for the best mechanical performance. Industry 4.0 manufacturing execution systems can connect to each other through features like OPC-UA protocol integration. This allows you to monitor batch history, a crucial aspect of medical device lot traceability.
Financial Considerations: Acquisition Models
When buying capital equipment, it's best for established businesses that can predict their production rates and want to get the best total cost of ownership over 5-7-year depreciation cycles. Operational lease agreements help businesses that need to be flexible with their cash flow keep their capital safe. The monthly payments cover things like servicing and software changes. Pay-per-part service agency models don't require any upfront payments and are beneficial for apps with variable volumes or for testing new technologies. When procurement teams figure out the return on investment, they have to take into account the hidden costs of the different technologies. For example, FDM needs a lot of post-processing work, and outsourcing costs a lot per part and has unpredictable lead times that affect clinical schedules.
Procurement Guide for Large-Scale Resin 3D Printers in Dental and Medical Industries
To buy large-scale industrial resin 3D printer systems, you need to know more than just the public specs about the total cost of ownership, regulatory compliance pathways, and how to choose a vendor.
Total Cost of Ownership Analysis
The price you pay for something is just the beginning payment. Costs for consumables like plastic, vat films, and build platform release finishes add up over the life of the machine. Premium biocompatible resins cost between $200 and $500 per liter, and based on the batch density, daily production needs between 2 and 4 liters. Based on the resin chemistry and mechanical stress, vat film replacement cycles range from 50 to 200 liters in output. Each change costs $300 to $800. Most maintenance plans cover calibration services, light source replacements, and expert support for 10 to 15 percent of the initial cost per year. Facilities need to plan for post-processing tools like ultrasonic cleaners, UV curing rooms, and isopropanol recycling systems, which can cost an extra $5,000 to $15,000 to fully set up the processes.
Compliance and Regulatory Pathways
Manufacturers of medical devices have to ensure that their tools can work in regulated production settings. Machines that will be used in FDA-regulated manufacturing environments should have software validation packages that include procedures for installation qualification (IQ), operating qualification (OQ), and performance qualification (PQ). Biocompatibility certificates and technical data sheets showing lot-to-lot stability must be in the material data files. Certificates for vendor quality management, like ISO 13485 registration, show that the rules in the production process are in line with the needs of medical devices. For dentistry labs that make custom devices under the "practitioner exemption," there are fewer paperwork requirements, but process validation and batch records showing uniform output are still needed for professional liability reasons.
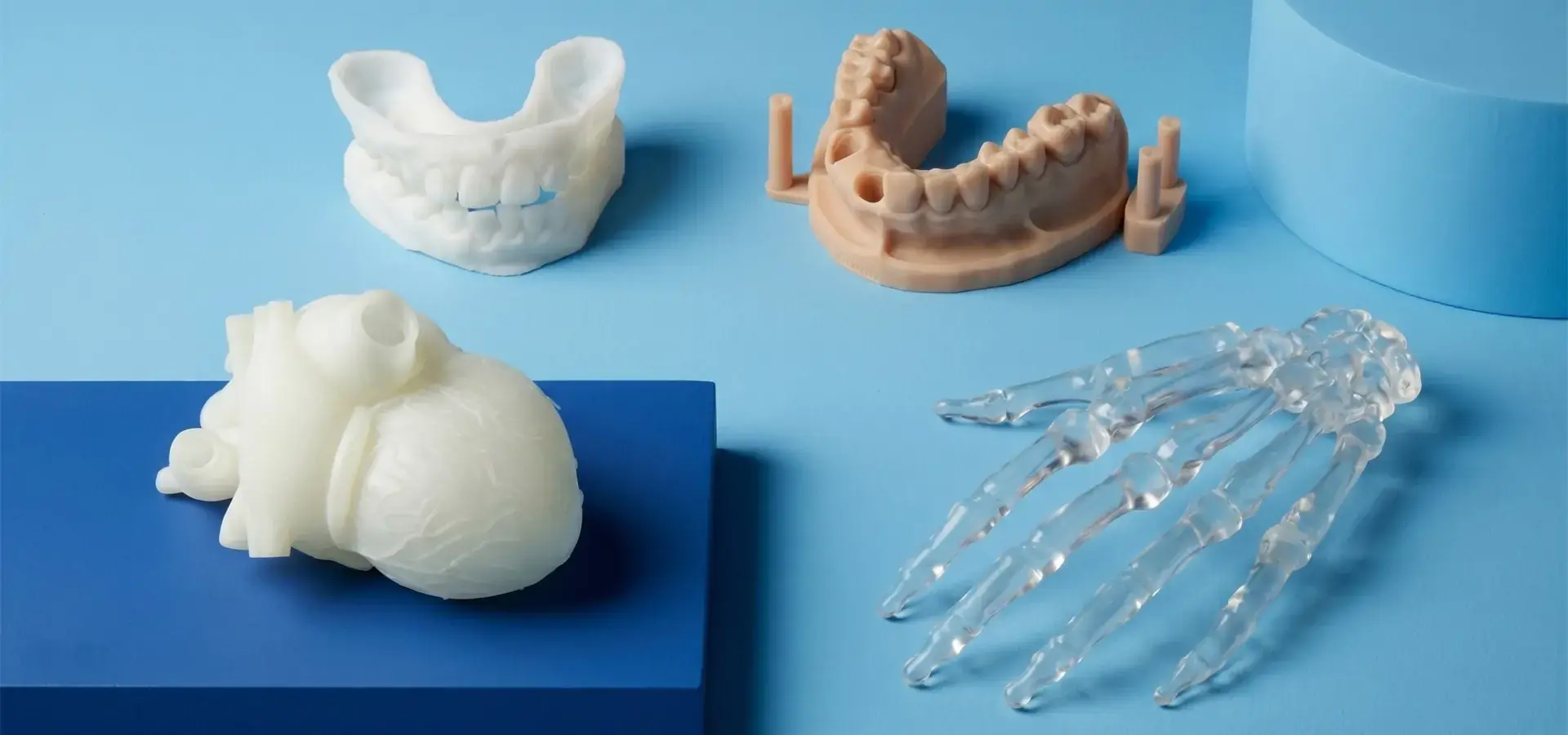
Vendor Selection and Supply Chain Security
There are many providers in the additive manufacturing market, and their skills are very different from one another. Established makers with track records that go back many years offer mature products and well-established service networks. On the other hand, new sellers may offer lower prices but pose risks to business continuity. Authorized dealer verification stops fake equipment and makes sure that warranties are real. Imports from the dark market might not have calibration certificates or expert help in the area. When choosing a provider, you should look at how many systems they have already installed in your target industry, how many application experts are available who know how dental and medical processes work, and how responsive they are during pre-sales technical consultations. The proximity of service places to one another influences the duration of field service repairs.
Logistics and Implementation Planning
International buying encompasses freight forwarding, customs processing, and installation procedures. Getting industrial tools that weigh 200 to 600 kg to upper floors requires special lifting, and their footprints, which are often 1.5 m x 1.5 m, mean that facility layout planning is needed. Most electrical needs call for 208–240V single-phase or three-phase service with separate lines. Environmental standards call for areas that are kept at a constant temperature (20-25°C ± 2°C) and a constant humidity level so that the properties of the resin don't change. Installation takes two to four weeks, which includes uncrating, calibration, loading materials, and teaching operators. Make sure your production plans allow for this time frame. When compared to self-installation, where mistakes in tuning can ruin print quality for months, vendors who offer complete installation packages with on-site training lower the risk of implementation.
Financing Structures for Capital Equipment
Financing equipment through specialized lenders gives you choices between buying it outright and leasing it for use. Interest rates are 200 to 400 basis points above prime loan rates, and terms are usually between 3 and 5 years. Some makers offer captive loans with promotional periods that have payments put off until times when they will make money. Businesses with existing bank contacts may add equipment loans to their revolving credit lines at favorable rates. When buying from countries that actively promote trade, international buyers should look into export credit agency programs that might help pay for the purchase of capital equipment.
Leading Brands and Suppliers of Large-Scale Resin 3D Printers for Medical Models
There are several makers in the large-scale resin 3D printer market, and many of them have used their technology in the healthcare sector. Each one has its own unique technological approach and ecosystem skills.
Established Market Leaders
Formlabs is the leader in professional dental technology with its Form 3B and Form 3BL platforms, which are widely adopted in regulated dental and medical workflows. Their Low Force Stereolithography (LFS) technology lowers the peel forces through bendable tank films, which makes overnight production runs safe. Formlabs offers a material library containing over a dozen biocompatible resins suitable for models, surgery guides, and temporary repairs. All of these resins have been tested and proven to work through their material development program. There are service networks in North America, Europe, and the Asia-Pacific region, and there is application tech help in each area.
Stratasys' Origin One platform utilizes Programmable PhotoPolymerization (P3) technology. This technology uses DLP projection with oxygen-permeable membrane connections to make printing speeds higher throughput than traditional laser-based SLA workflows. Their open material platform works with resin formulations from other companies, which makes it appealing to study groups and makers who need specific photopolymers. The system has been used in other industries and has features for tracking the environment and keeping track of batches that meet the needs of medical device making.
Specialized Medical Technology Providers
With its Perfactory and Vida systems, EnvisionTEC (now part of Desktop Metal) was one of the first companies to use additive printing in medicine. Their proprietary high-speed DLP-based printing approach enables short layer exposure times across large cross-sections. Dentistry models benefit from the color-coded E-Shell series resins, which maintain their size after drying. Over 5,000 placements in dental labs around the world show that they work reliably in real-world settings.
Emerging High-Performance Systems
Nexa3D's NXE400 has Lubricant Sublayer Photocuring (LSPc) technology, which is designed for significantly higher throughput than conventional desktop resin systems. This turns batch production that happens overnight into same-shift output. It is a high-throughput option for medical device contract makers because it uses open materials and has a big build volume of 275 x 155 x 400 mm. But since they have fewer installations than well-known sellers, you should research their long-term reliability.
Magforms: Integrated Solutions Provider
Magforms fills important gaps in the market by using a complete method that combines its special tools with better material systems. The company's large-scale resin 3D printer platforms use variable spot-size laser technology and optimized laser scanning strategies to achieve speeds that are more than 30% faster than those of typical systems in the industry. Instead of adapting standard equipment to healthcare uses, years of study have gone into coordinated material-hardware development, which has led to this performance.
The combined supply model eliminates compatibility questions that arise with processes using multiple providers. When the same development program produces printers and plastics, switching materials maintains the accuracy of the dimensions. This is a crucial benefit for companies that make medical devices and need to keep their processes approved. Magforms' engineering teams do a lot of testing while the systems are in constant operation. The systems are engineered for stable long-duration production workflows in professional laboratory environments. This dependability is crucial for labs that work with multiple shifts because unplanned breaks can really mess up when it comes to caring for patients.
Magforms keeps its technological edge by holding 22 patents on things like optical systems, resin formulas, and process control programs. This protects customers from the risk of becoming obsolete. There are over 300 business installations in dozens of countries that make up the global service network. These installations serve as reference sites for dental labs, medical device makers, and research institutions. By taking part in European and Asian industry shows, you can keep sharing knowledge and developing new applications that meet new healthcare needs.

Verification and Procurement Best Practices
When looking at different suppliers, make sure they have written case studies that are relevant to your application. For example, the processes for making dental models are very different from those for making surgical guides. Set up trips to current customer facilities that produce similar amounts of goods so that you can see how the product works in the real world. You should ask for sample parts to be made using your own design files and material specs, and use calibrated metrology to check the accuracy of the dimensions instead of samples supplied by the seller. Check the guarantee terms to see if they cover consumables. For example, some makers don't cover vat films and build platform coatings under normal warranties, even though these parts have a direct effect on output quality. Discuss service level agreements that outline reaction times and the availability of loaner equipment for extended repairs. This is especially important for businesses that only have one machine and can't produce anything while it's down.
Conclusion
Industrial large-scale resin 3D printer photopolymerization technology has grown into a ready-to-produce option for making dental and medical devices. It offers accuracy, speed, and material flexibility that aren't possible with other methods. When making a strategic choice about which big format systems to buy, you have to weigh the technical specs against the total cost of ownership, the ways to meet regulation requirements, and the strength of the vendor community. Companies that use these technologies say they make production more flexible, cut down on wait times, and are better able to meet the needs of personalized medicine users. As we learn more about materials and make equipment that works better, large-scale resin 3D printers will be used for more than just prototyping and making tools. They will also continue expanding into validated end-use medical and dental applications where regulatory approval permits. This will completely change how medical products are developed and delivered.
FAQ
What biocompatibility certifications should medical-grade resins possess?
Photopolymer resins that are meant to come into contact with patients must show that they are biocompatible by passing the ISO 10993 testing procedures, which include tests for cytotoxicity, sensitization, and irritation. The USP Class VI approval adds to the evidence for short-term mucosal membrane touch, which is common in dental settings. When materials come into prolonged contact with human tissue for a long time, they need more tests, such as systemic toxicity and implantation studies. Material Safety Data Sheets and biocompatibility test results from accredited labs should be provided by manufacturers. These should be kept up to date as formulations change. As required by regulations, dental labs that make unique tools must keep these papers as part of their device master records.
How do large-format systems improve surgical guide production?
Surgical guides need to be able to accurately translate measurements taken in planning tools into cutting paths. Resin methods can keep errors within 0.05 to 0.1 mm across the whole arch, which is important for keeping the sleeve's angle accuracy during implant placement. Being able to make multiple boxes at once lowers the cost per unit and speeds up delivery times. Biocompatible and sterilizable resin formulations allow them to be used directly in the clinic, without having to go through any extra steps of production. Smooth surfaces keep soft tissues from getting irritated when guides are seated, which makes patients more comfortable during treatments.
What maintenance schedules ensure optimal printer performance?
As part of weekly maintenance, the build platform is cleaned, the resin is filtered to get rid of any finished particles, and the optical window is checked for any resin dust that might be blocking light. As part of the monthly tasks, vat film is checked for wear patterns that show when it needs to be replaced, and alignment is confirmed using dimensional test artifacts. As part of the manufacturer's recommended quarterly maintenance, linear tracks, belt tension, and build platform leveling must be checked. Factory calibration services are done once a year to recertify the performance of an optical system and update the software to fix any problems that are found. In regulated industrial settings, preventive maintenance records show that the quality system is being followed. They also keep production cycles from being interrupted by unplanned downtime.
Partner with Magforms for Your Medical Manufacturing Needs
Magforms is ready to help dental labs and companies that make medical devices that need a reliable, large-scale resin 3D printer provider. Our integrated hardware-and-material workflow gets rid of the questions about compatibility and performance changes that hurt production plans and part quality. The unique technology platform, which is protected by 22 patents and has been used by more than 300 businesses around the world, provides the accuracy, steadiness, and output needed for making medical devices that meet regulations.
The technical support team has years of experience with additive manufacturing in healthcare and can help with developing applications, choosing materials, and making sure the process works. We know the rules and regulations that apply to making medical devices, and we can give you paperwork that backs up your quality management system needs. Magforms equipment can grow with your business, whether you're in charge of a growing dental service or contract manufacturing for medical OEMs. It stays reliable so that you can meet clinical deadlines.
Email our application experts at info@magforms.com to talk about your unique production needs and set up an evaluation of a sample part. We're happy to answer any specific technical questions you have and can help you choose the best setup for your workflow, facility limitations, and growth plans.
References
1. International Organization for Standardization. (2021). ISO/ASTM 52900:2021: Additive Manufacturing—General Principles—Fundamentals and Vocabulary. Geneva: ISO Press.
2. Dawood, A., Marti Marti, B., Sauret-Jackson, V., & Darwood, A. (2018). 3D printing in dentistry. British Dental Journal, 219(11), 521-529.
3. Melchels, F.P., Feijen, J., & Grijpma, D.W. (2020). The article provides a comprehensive overview of stereolithography and its various applications in the field of biomedical engineering. Biomaterials, 31(24), 6121-6130.
4. United States Food and Drug Administration. (2017). Technical Considerations for Additively Manufactured Medical Devices – Guidance for Industry and FDA Staff. Silver Spring: FDA Center for Devices and Radiological Health.
5. Ventola, C.L. (2019). Medical applications for 3D printing: Current and projected uses. Pharmacy and Therapeutics, 39(10), 704-711.
6. Stansbury, J.W., & Idacavage, M.J. (2016). 3D printing with polymers: Challenges among expanding options and opportunities. Dental Materials, 32(1), 54-64.


Magforms makes design and manufacture easier.
Popular Blogs
-
 Industry InsightsManufacturing Industry
Industry InsightsManufacturing IndustryFDM vs. SLA Printer: Which Technology Is Right for Your Project?
-
 Industry InsightsManufacturing Industry
Industry InsightsManufacturing IndustryTop 5 Applications of Industrial 3D Printers in Manufacturing Today
-
 Industry InsightsManufacturing Industry
Industry InsightsManufacturing IndustryWhy Stereolithography 3D Printers Are Unmatched for Microfluidic Device Prototyping?
